 In the meantime, Teva’s success is a testament to its dedication to its patients. This is a sign of a thriving pharmaceutical industry. The approval means that Teva has 180-days’ market exclusivity, and is not subject to generic competition. Meanwhile, the company is still awaiting the FDA’s approval for the generic version of Adderall. However, the company has been left in the dust by generic companies. Its main rival, Novartis, uses the Sandoz generic arm to protect its brand. Teva is the largest producer of the drug in the world, and it has the most patents. It said that if the FDA had approved counterfeit drugs, it would be able to stop them in their tracks. The company also issued a warning regarding the sale of fake Adderall online. The legitimate tablets contain dextroamphetamine sulfate, aspartate, and aspartate. A counterfeit tablet contains tramadol and acetaminophen, which are both used to treat acute pain. The new tablet contains a mixture of amphetamine salts. In the meantime, the manufacturer of the generic Adderall XR capsules has begun shipping them in the United States. While Teva is committed to the safety of its consumers, the FDA is a key factor in determining the safety of the drug. Despite the warning, a number of pharmacists have expressed concern about the counterfeits. 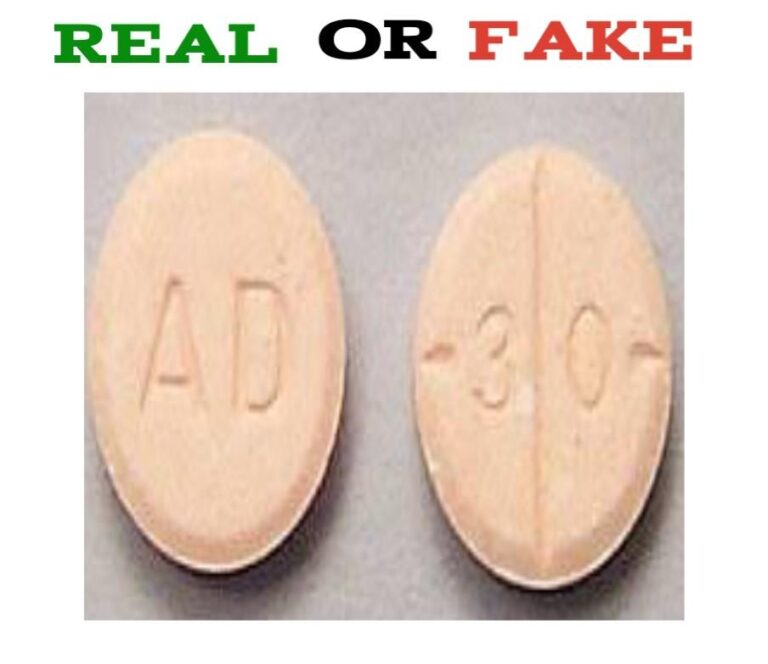
The FDA has also approved generic versions of Yaz Tablets, which are used to treat anorexia. The generic Adderall XR capsules are approved by the FDA to treat hyperactivity. When in doubt, they can order generic Adderall. This means pharmacists must maintain strict records of the amount they dispense. Unlike its counterfeit counterparts, Adderall is classified as a controlled substance, meaning it must be dispensed under special regulations. Teva has also released generic Adderall XR capsules, which are approved for the treatment of attention deficit hyperactivity disorder and narcolepsy. There is also a risk of a contaminated pill if you buy counterfeit Adderall from an online drugstore. 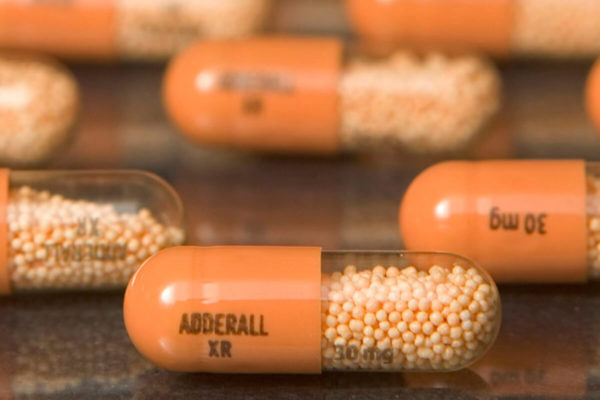 
The FDA considers these pills to be controlled substances, so pharmacists must follow strict guidelines in dispensing them to patients. Food and Drug Administration (FDA) has issued a warning about counterfeit Adderall 30 mg tablets, which are commonly used to treat ADHD and narcolepsy. The tablets themselves are white in color, with no markings.Īs of September 2013, the U.S. #How to tell if adderall is fake code#While it is not easy to identify a counterfeit Adderall product, Teva’s generic Adderall tablets are easily identified by their round, orange/peach color and are marked with “dp” and “30.” They are packaged in 100-count bottles and have a National Drug Code (NDC) of 0555-0768-02. Because of these issues, Adderall is currently on the FDA’s drug shortage list. In addition, counterfeit products often contain acetaminophen, which are common in acute pain medicines. The original formulation of Adderall contains dextroamphetamine sulfate, dextroamphetamine aspartate, amphetamine sulfate, and tramadol. The FDA recently reported that counterfeit Adderall 30 mg tablets contained the wrong active ingredients. 
0 Comments
Leave a Reply. |
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |
 RSS Feed
RSS Feed
